Elevate Transparency Disclosure Management with Compliant and Effective Solutions
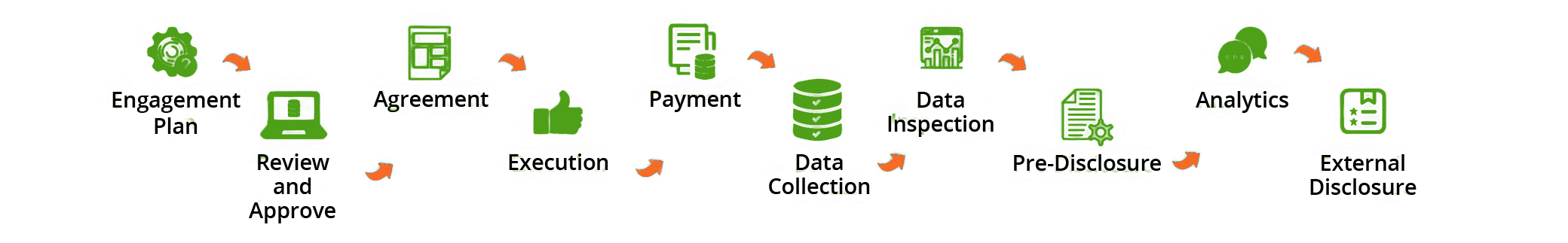
Challenges in Transparency Disclosure

Complex global laws and self-regulatory codes

Data completeness and accuracy

Varied reporting templates and requirements

Pre-disclosure & consent requirements

Master data related challenges
Data in different sources e.g., different ERP systems

Analytics and Impact Management

Support structure to manage operations
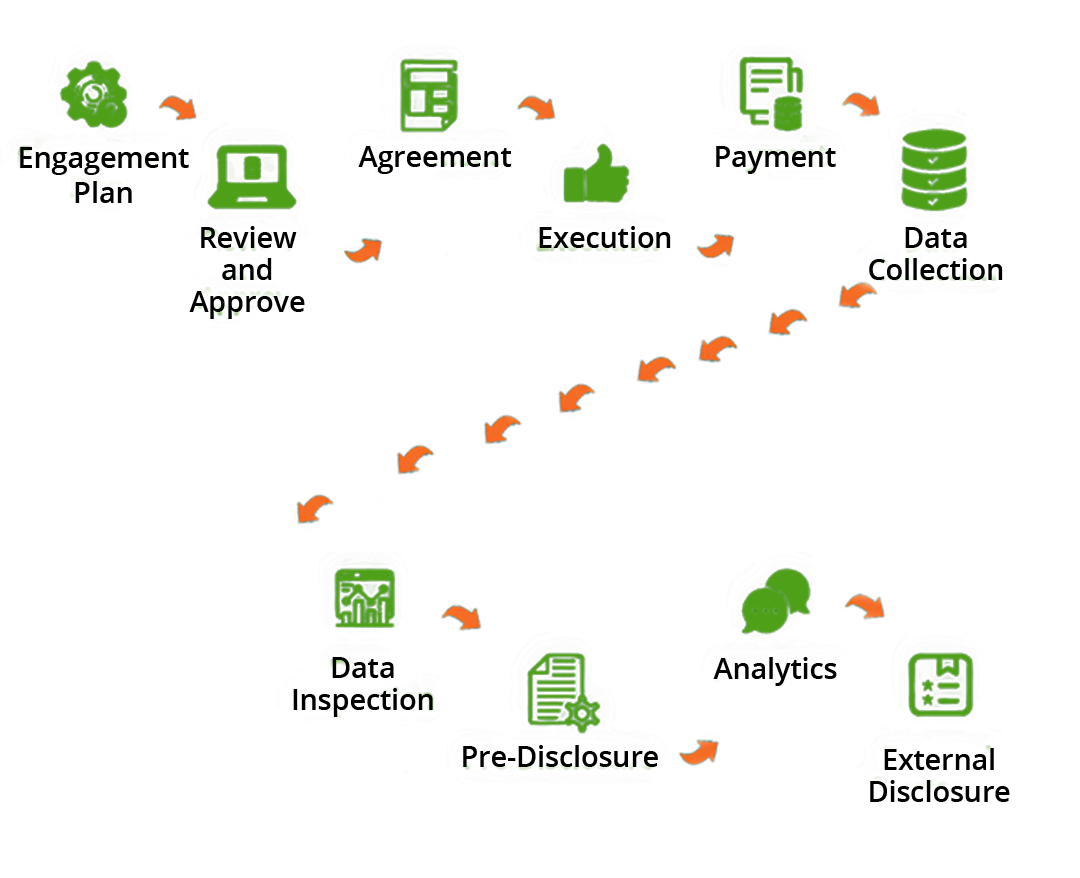
How Can Eunomia Help You Overcome These Challenges
Follow the Money
By building a comprehensive methodology to allow the identification of reportable Transfers of Value based on payments posted in the ERP system or any other finance systems. Thus, reducing the risk of relying on manual files or other data sources.
Simplify the Complex Legal Landscape
We have all the expertise to manage all the contract templates to include the relevant verbatim for consent, data privacy etc. We can support with providing templates and structure to manage pre-disclosure and consent requirements as needed
Reporting Requirements and Templates
We understand the complex landscape of law and self-regulatory requirements around disclosure and are up-to-date with any changes. We have a repository of all the country-specific templates required for disclosure and can also support with building templates, where needed
End-to-End Process Build and Enablement
We can support with the build of a transparency process, provide operational support for existing processes and provide best practice around how to manage the process effectively to achieve best results
Timely and Accurate Analytics of HCP/O Spend
Through our service model we can provide a timely reporting of spend with HCPs, HCOs, etc. enabling effective compliance spend analytics and monitoring
Global or Local Solution
Whether you are looking to build a global process that can be cascaded locally, or build a localized process to meet a specific country requirement, we can help

Our Role in Sustainability Reporting
At Eunomia Pharma Services, we understand that sustainability is no longer optional—it's integral to compliance. Whether you're reporting for the first time or enhancing your current ESG disclosures, our compliance professionals can help you navigate complexity with confidence. We help you embed sustainability into your compliance framework by supporting accurate, consistent, and audit-ready ESG reporting. We can support the drafting and review of sustainability and ESG reports.
- Ensuring data is structured, verifiable, and aligned with reporting standards.
Book a Consultation
FAQs
What are Transparency Disclosure Services?
Eunomia Pharma Services Transparency and disclosure services support pharmaceutical, biotech, and medical device companies in managing and disclosing transfers of value made to external stakeholders, in line with applicable laws, regulations, and industry codes.
These requirements arise under national “Sunshine” laws in several EU countries, as well as under self-regulatory frameworks such as EFPIA and ABPI, with disclosure expectations continuing to evolve across Europe.
Our services cover the end-to-end transparency process, including data cleaning, validation and reconciliation, aggregation, population of country-specific disclosure templates, and submission on the relevant national platforms or portals—supporting both mandatory statutory disclosures and voluntary code-based reporting in a consistent and compliant manner.
Who needs transparency and disclosure support?
Any pharmaceutical, biotech, or medical device organisation that makes transfers of value to external stakeholders, such as healthcare professionals (HCPs) or healthcare organisations (HCOs), requires transparency support.
Transparency support is relevant for companies operating in Europe, the UK, and other regulated markets where disclosure is required under national Sunshine laws or expected under industry self-regulatory codes, as well as for organisations choosing to implement voluntary transparency frameworks to strengthen governance and public trust.
What activities fall under transparency reporting?
Transparency reporting covers transfers of value made to healthcare professionals (HCPs) and healthcare organisations (HCOs), including fees for service and consultancy (such as advisory boards and speaker engagements), sponsorship of events, contributions to event-related costs (including registration fees), travel and accommodation, and grants or donations.
The scope, categorisation, and disclosure format may vary by country based on local laws and regulations. Our approach ensures activities are assessed and disclosed in line with applicable local transparency rules while maintaining consistency across markets.
Which regulations and codes do you support?
We provide global transparency and disclosure support aligned with applicable laws and industry codes across multiple jurisdictions. Examples include the US, France, Portugal, and other EU countries, in line with emerging sunshine laws, each with distinct scope, formats, and submission platforms. Our services are designed to support both mandatory statutory disclosures and voluntary code-based reporting, while ensuring local legal compliance within a consistent global governance framework.
How do you support the transparency lifecycle?
We provide end-to-end support, including transparency framework design, data capture models, Fair Market Value alignment, reconciliation and validation processes, governance documentation, disclosure preparation, and audit readiness support, ensuring transparency is operationally sustainable, not just a year-end exercise.
Can transparency processes be customised to our organisation?
Yes. We design and operate transparency frameworks tailored to your organisational structure, engagement models, systems landscape, and geographic footprint. Depending on your needs, we can support specific components of the transparency lifecycle or fully outsource your end-to-end transparency and disclosure activities, acting as an extension of your internal compliance and finance teams.
How do you ensure data accuracy and audit readiness?
We apply structured validation checks, reconciliation logic, and documented governance controls throughout the transparency process. This ensures disclosures are defensible, traceable, and aligned with regulatory expectations, supporting both internal audits and external inspections.
Can you help us convert risk assessment findings into action plans?
Absolutely. Beyond operational support, we help organisations strengthen transparency governance through risk assessments, control design, SOP development, training, and role clarity—ensuring accountability across compliance, finance, and business teams.
How does transparency link to broader healthcare compliance?
Transparency is a critical pillar of healthcare compliance. Effective transparency processes reinforce ethical engagement, support anti-bribery and corruption controls, and provide regulators with confidence that interactions with HCPs and HCOs are appropriate, proportionate, and well-governed.
How do we get started with Eunomia Pharma Services?
You can contact us to discuss your transparency challenges and operating model. We’ll assess your current state and design a pragmatic, compliant transparency solution tailored to your organisation and markets.
UK regulations for drug company data sharing with the public.
Are there services that track pharmaceutical companies’ transparency disclosures?
How do pharmaceutical transparency disclosures impact drug pricing?
Understanding the impact of pharmaceutical marketing spend disclosure.
What is the ABPI Code of Practice and its relevance to transparency?
How to manage EFPIA disclosure reporting?
Eunomia Pharma Services supports EFPIA disclosure by helping pharmaceutical companies design, implement, and operate compliant, end-to-end transparency frameworks aligned with the European Federation of Pharmaceutical Industries and Associations Code. This includes developing clear governance models, defining roles across compliance, finance, medical, and commercial teams, and building robust SOPs covering Transfers of Value (ToV) identification, data capture, validation, consent management, and disclosure reporting. The focus is on creating practical, inspection-ready processes that work seamlessly across global and local teams.
A core strength lies in data governance and operational execution. Eunomia supports companies in standardising HCP/HCO master data, integrating multiple source systems (CRM, ERP, event and grants tools), and establishing a central transparency reporting layer. This ensures accurate data capture at source, reduces manual errors, and enables efficient reconciliation between finance and disclosure systems. At the same time, Eunomia ensures alignment with the General Data Protection Regulation, including consent management and handling of country-specific disclosure requirements, which vary significantly across EFPIA markets.
In addition, Eunomia provides hands-on support through risk assessments, reporting, and managed services. This includes conducting transparency health checks, preparing companies for audits and inspections, identifying and remediating gaps, and supporting end-to-end disclosure cycles—from data validation through to report generation and publication. For organisations with lean internal teams, Eunomia can act as an extension of the compliance function, delivering scalable, accurate, and timely EFPIA disclosures across multiple countries.
Sunshine Act compliance support services
Eunomia Pharma Services supports pharmaceutical companies with end-to-end Sunshine Act and global transparency compliance by combining regulatory expertise with practical implementation. This includes interpreting requirements under frameworks such as the Physician Payments Sunshine Act and the European Federation of Pharmaceutical Industries and Associations Code, and translating them into clear, workable compliance frameworks. Eunomia helps organisations design governance models, define roles and responsibilities, and build robust SOPs that ensure accurate identification, capture, and reporting of Transfers of Value (ToVs).
A key strength lies in operational execution and data governance. Eunomia works across systems (CRM, finance, events, and grants) to standardise data capture, improve data quality at source, and create integrated transparency reporting processes. This includes reconciliation between finance and disclosure systems, validation of data, and preparation of submission-ready reports. At the same time, Eunomia ensures alignment with the General Data Protection Regulation, supporting consent management, privacy considerations, and country-specific requirements—particularly important in markets where disclosure rules vary.
Beyond framework design, Eunomia provides hands-on support through risk assessments, training, and managed services, acting as an extension of internal compliance teams. This includes conducting health checks, preparing companies for audits and inspections, identifying gaps, and implementing remediation
ABPI transparency reporting requirements
Eunomia Pharma Services helps pharmaceutical companies meet their ABPI transparency reporting obligations by building robust processes and systems to manage the entire disclosure lifecycle. They can assist in aggregating and validating payments or transfers of value to healthcare professionals (HCPs), healthcare organisations (HCOs), patient organisations and other external stakeholders so that data captured from finance and engagement systems is complete, accurate and aligned with ABPI’s requirements. Their support includes designing workflows and tools that handle pre‑disclosure steps, such as consent and data cleansing, and producing audit‑ready transparency reports that comply with ABPI, EFPIA and other international codes, thereby streamlining compliance and reducing manual work.
In addition to technical reporting support, Eunomia also offers policy, SOP and compliance framework development that incorporates ABPI’s transparency standards into a company’s governance documentation. This ensures internal practices are consistently aligned with regulatory expectations and allows organisations to maintain a defensible approach to disclosure on an ongoing basis. Their services can be tailored globally or locally, giving companies the flexibility to implement ABPI‑aligned reporting processes that work across jurisdictions and evolve with changing regulatory landscapes.
Difference between EFPIA disclosure requirements and US Sunshine Act
Feature EFPIA Disclosure (Europe) US Sunshine Act (Open Payments) Scope Covers transfers of value (ToVs) to healthcare professionals (HCPs) and healthcare organizations (HCOs) across EFPIA member countries. Covers payments or transfers of value to physicians (MDs, DOs) and teaching hospitals. Geographic Coverage 37 European countries where EFPIA code applies. United States only. Reporting Entities Pharmaceutical companies that are EFPIA members. Applicable manufacturers of drugs, devices, and biologics regulated by the FDA. Data Types Reported Fees for services, consultancy, travel & accommodation, grants, donations, sponsorships, etc. Consulting fees, honoraria, gifts, food, travel, research payments, ownership interests. Consent Requirement HCP consent is required in some countries for individual-level disclosure; otherwise, aggregate disclosure is allowed. No consent needed; all payments must be reported at individual physician level. Disclosure Frequency Annually (varies by country; usually following calendar year). Annually (data reported to CMS by March 31; public by June 30). Public Access Published on company websites or national databases; not centralized across EU. Centralized public database (CMS Open Payments) accessible online. Penalties for Non-Compliance National-level enforcement; varies by country (can include reputational risk and fines). Civil monetary penalties and potential exclusion from federal healthcare programs. Level of Detail May be aggregated or individual-level depending on country and consent. Individual-level reporting for each physician and teaching hospital. Purpose Promote transparency in pharma-HCP interactions, align with EFPIA Code of Practice. Promote transparency in healthcare financial relationships, prevent conflicts of interest.
